In this article, I will explain the concept of electromotive force or EMF. EMF is nothing but the amount of energy supplied by a source to a unit charge to complete its path in a circuit. Electromotive force or EMF is not a force, but it is an energy given to electric charge by the source of energy. In this article, I will cover all the concepts of EMF. So, let’s begin with the basic definition of electromotive force (EMF).

What is EMF?
EMF stands for Electromotive Force. It is defined as the amount of energy the source like a battery gives to each coulomb of charge.
EMF is always defined as a source of energy like a battery, cell, or generator, and it cannot be specified for a load device like a resistor.
There are various methods of producing EMF, such as in a battery, the EMF is produced by chemical reactions, in a generator, the EMF is produced by the magnetic field, in a solar PV cell, the EMF is produced by light, and so on.
In general, the EMF is denoted by the symbol “E” or “e”. Where, “E” is used to denote a constant valued electromotive force like EMF of a battery, whereas the letter “e” is used to denote a time-varying emf, like emf produced by an AC generator.
EMF Formula
By the definition of EMF, the electromotive force or EMF is the energy supplied per unit charge by the source of energy.
Hence, mathematically, it can be expressed as,
$$E=\frac{\text{Energy }(W)}{\text{Charge }(Q)}$$
This is the fundamental expression of EMF. Where, energy is measured in Joules and charge is measured in Coulombs.
Unit of EMF
The unit of emf can be derived from its formula given above.
$$\text{Unit of }E=\frac{\text{Joule }(J)}{\text{Coulomb }(C)}$$
Hence, the unit of EMF is Joule per Coulomb (J/C). This is also known as Volt (V).
1 V = 1 J/C
EMF of a Cell
The electric circuit model of a cell is shown in the following figure.
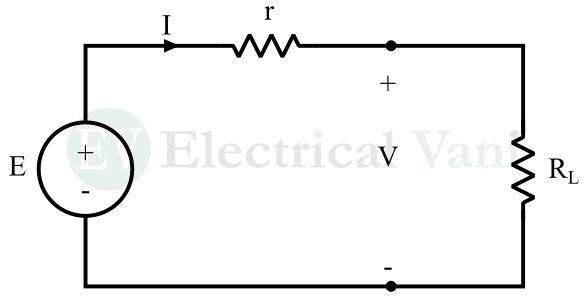
In this circuit, E is the EMF of the cell, r is the internal resistance of the cell, and V is the terminal voltage of the cell.
When a load resistor RL is connected across the terminals of the cell, an electric current starts flowing in the circuit.
Writing KVL equation for the circuit, we get,
V = E – Ir
Therefore, the EMF of the cell will be given by,
E = V + Ir
Hence, the EMF of a cell is the sum of terminal voltage and the voltage drop across the internal resistance of the cell.
Difference between EMF and Potential Difference
From the above discussion, we can see that the unit of EMF is volt. As we know, volt is also the unit of voltage or potential difference. Hence, it is crucial to understand the difference between emf and potential difference. For this, let us consider an electric circuit containing a battery, and two resistors connected in series as shown below.
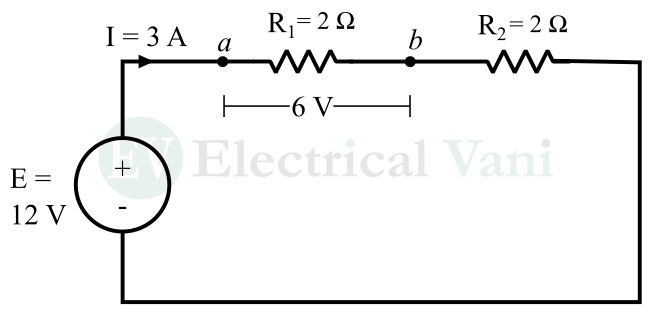
In this circuit, the battery has an EMF of 12 V, which means it supplies 12 Joules of energy to each coulomb of charge.
On the other hand, the potential difference is defined as the amount of energy used by each coulomb of charge to move from one point to another.
In this case, consider one coulomb of charge moving from points a to b. We are given that the potential difference between points a and b is equal to 6 V. It means that each coulomb of charge requires an energy of 6 joules to move from point a to b.
Therefore, EMF is the total energy that the battery gives to each coulomb of charge, while the potential difference between two points is the energy required by the charge to move from one point to another.
Relationship between EMF, Voltage, and Current
In an electric circuit, EMF, voltage, and electric current are the three fundamental electrical quantities.
These three electrical quantities are related to each other as follows:
A source of energy produces and maintains an EMF. This EMF of the source creates a potential difference between the two points. The potential difference causes an electric current to flow between those two points from the point of high potential to the point of low potential.
Numerical Examples Based on EMF
Example (1) – A battery supplies an energy of 500 joules to each coulomb of charge. What will be the EMF of the battery?
Solution – Given data,
Energy (W) = 500 Joules
Charge (Q) = 1 Coulomb
The EMF of the battery is,
$$E=\frac{W}{Q}=\frac{500}{1}=500 \text{Volts}$$
Example (2) – The internal resistance of a cell is 0.12 Ω. If the terminal voltage of the cell is 2.25 Volts. Then, calculate the EMF of the cell, if the cell produces an electric current of 2 A when connected in a circuit.
Solution – Given data,
Internal resistance of cell, r = 0.12 Ω
Terminal voltage, V = 2.25 V
Circuit current, I = 2 A
The EMF of the cell will be,
E = V + Ir
Substituting the values in the formula of emf, we get,
E = 2.25 + (2 × 0.12)
E = 2.49 Volts
Conclusion
In conclusion, EMF (Electromotive Force) is the amount of energy given by a source like a cell to per unit charge. It is the most elementary electrical quantity in a circuit which is responsible for the existence of potential difference and electric current. The EMF is measured in volt, however, it is different from the potential difference or voltage. EMF is always defined for a source element like a generator, battery, cell, etc. It cannot be specified for a load device like a resistor or a motor.
In this article, I have explained the concept of EMF in detail. If you have any queries related to this topic, please let me know in the comment section.